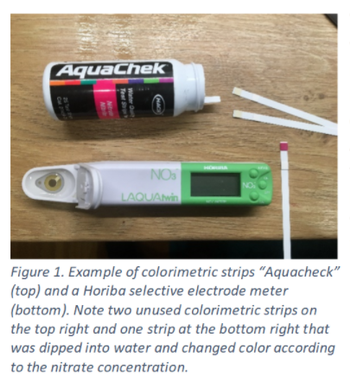
How To Measure Nitrate Concentration

Featured Story
By Gerry Spinelli, Production Horticulture Advisor with the University of California Cooperative Extension
If you inject fertilizer into your irrigation system, you probably have a target concentration of nitrate that you are trying to achieve. Is your injector set at the right dilution ratio? Did your irrigator mix fertilizer correctly into the fertilizer stock solution? The best way to answer these questions is to measure nitrate in the liquid feed that is distributed to your plants.
You can take a sample from your liquid feed and take it to a water quality lab for measurement, or you can estimate nitrate concentration with a hand-held selective electrode meter (manufacturers include Horiba and Hanna) or with colorimetric strip tests like the Merck MQuant or the Hach Aquachek. These methods are not very accurate but have the advantage of being quick and affordable. The main problem that you will encounter when using these tools is that nitrate concentration is expressed in different units and it’s easy to get confused.
Nitrate concentration in water is typically measured in parts per million (ppm) or milligrams per liter (mg/L). Since one liter of water weighs 1000 grams or one million milligrams, stating milligrams per liter is like saying milligrams per million milligrams, so milligrams per liter and part per millions are (almost) the same. At the concentrations used in horticulture, ppm and mg/L are interchangeably used.
It gets more complicated when we start talking about nitrate. You will encounter two units when dealing with nitrate: ppm of nitrate (NO3-) and ppm of nitrate-nitrogen (NO3--N). The latter can also be called nitrate-N, nitrate as nitrogen or NO3--N. The difference between the two units is that nitrate tells you the weight of the whole nitrate molecule (that contains one nitrogen and three oxygens), while nitrate-nitrogen tells you only the weight of the nitrate within that molecule. So, the concentration in units of nitrate-nitrogen will always show a smaller number than the same concentration expressed in units of nitrate.
The way we can keep it straight in our head is by thinking about a chicken. There is the weight of the chicken and there is the weight of the chicken-meat, chicken as meat or chicken-M. The chicken-meat always has smaller weight than the whole chicken since it doesn’t include the weight of bones, skins, etc. Similarly, nitrate-nitrogen always has smaller values since it doesn’t consider the weight of oxygen within the molecule.
The conversion factor is 4.43, meaning that if you have 1 mg/L of nitrate-nitrogen, just multiply it by 4.43, which would give you 4.43 mg/L of nitrate. This is like saying that 1 lb of chicken meat equals 4.43 lbs of whole chicken, including bones and skins. If you are converting from nitrate to nitrate-nitrogen, then divide it by 4.43, so, 1 mg/L of nitrate which equals to
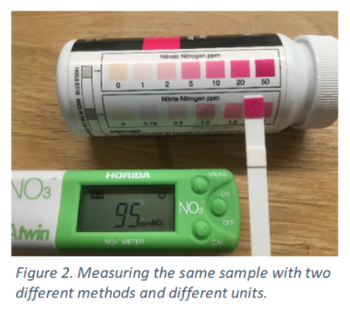
0.226 mg/L of nitrate-nitrogen. For example, in the image below, for a water sample the Horiba meter display shows 95 ppm of nitrate. For the same water sample, the Aquachek colorimetric strips gives a color corresponding to between 20 and 50 ppm of nitrate-nitrogen on the colorimetric scale on the label of the white bottle. The readings from these two instruments are essentially the same, since 95 ÷ 4.43 = 21.
Another example, say that your local Cooperative Extension advisor recommended a concentration of 50 mg/L of NO3--N for your liquid feed to grow Ligustrum. You take a sample at your dripline and your Horiba meter says 220 ppm of NO3-. To convert 50 mg/L of NO3--N to NO3- , we multiply by 4.43 as follows: 50 x 4.43 = 221 mg/L NO3--N so your sample measuring 220 ppm of NO3- is very close to the recommendations. Remember, ppm and mg/L are the same!
I encourage you to get familiar with this units, because measuring nitrate concentration is the best way to manage your nitrogen application; this will prove useful to save on fertilizer applications and to meet water quality regulations.
For questions or comments, please contact Gerry Spinelli, Production Horticulture Advisor with the University of California Cooperative Extension at (530) 304 3738 or email at gspinelli@ucdavis.edu